 
The solubility of these compound is governed by _Ī) Lattice energy: Larger the lattice energy, the lesser is the solubility.Į.g., sulphates and phosphates of Ba & Sr are insoluble in water due to high lattice energy.ī) Heat of hydration: Larger the heat of hydration, the more is the solubility of ionic compound.Į.g., AlCl 3, though covalent in nature is soluble in water due to high value of heat of hydration. The solubility of ionic compounds decreases with increase in covalent character of ionic compounds. Iv.They are generally soluble in polar solvents, having high value of dielectric constant but insoluble in non-polar solvents due to low dielectric constant. Ionic compounds have high melting points and boiling points due to powerful electrostatic force between ions.įor More Chemistry Doubts visit main page of Physics wallah. Ii. The crystalline ionic compounds have well defined crystal structure or crystal lattice. I. These compounds usually exists in the solid state. The important properties of ionic compounds are _ d.All ionic solids are similar in density.What are the properties of an ionic compound?.c.Molecular solids do not dissolve easily in water.b.Network solids are generally not soluble in water.a.Metallic solids have a high melting point.Which statement is true about the properties of solids? c.whose electrons are held with metallic bonds.b.whose electrons are held with covalent bonds.a.whose electrons are held with ionic bonds.To conduct energy most efficiently and effectively, she should use material She has four solids available, each made with different materials. It is possible to tell the difference between a solid with a crystalline structure and one with an amorphous structure just by looking at it.Īn engineer is designing an electrical system and is looking for a material to transmit energy. _ solids are known for their ability to be flattened into a sheet, stretched into a wire, and to conduct energy well. These differences affect how a solid conducts heat and electricity, its density, and The elements that make up the solids also differ. 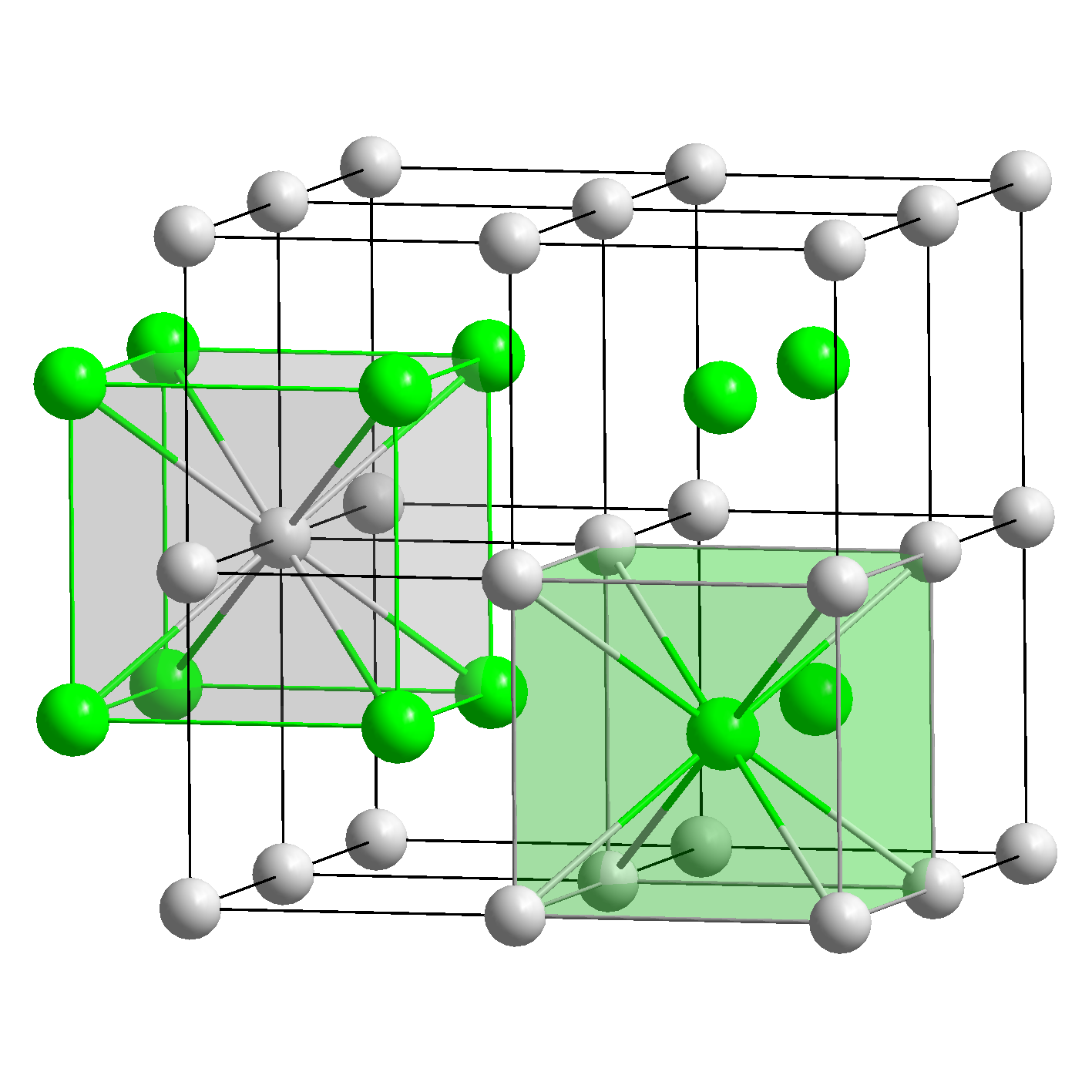
d.Crystalline solids all share a lattice structure, but the bonds that hold them together at the atomic level differ.c.Crystalline solids all share a lattice structure, but behave differently under similar conditions.These elements affect the way the solid conducts heat and electricity. b.Crystalline solids all share a lattice structure and the same types of bonds, but are composed of different elements.Chemists use the groups to organize the solids by density. a.Crystalline solids all share a lattice structure, but have different densities.Which explanation below will BEST help him begin to understand why chemists might have these groups? d.amorphous solids always behave consistently and uniformly.Ī friend in your chemistry class is struggling to understand why crystalline solids are grouped into four main types: network, molecular, ionic, and metallic.c.crystalline solids break unpredictably and can produce curved fragments. 
b.amorphous solids have a lattice structure.a.crystalline solids have a precise melting point.One major difference between crystalline and amorphous solids is that d.All solids have a lattice structure at the atomic level.c.All solids maintain a defined shape and size if conditions remain constant.b.All solids have a crystalline structure.a.Solids maintain a defined shape and size under all conditions.Which of the following is true of solids? In amorphous solids, the atoms or molecules are held together in a completely random formation. Understanding Scientific Journals and Articles.Using Graphs and Visual Data in Science.Scientists and the Scientific Community. 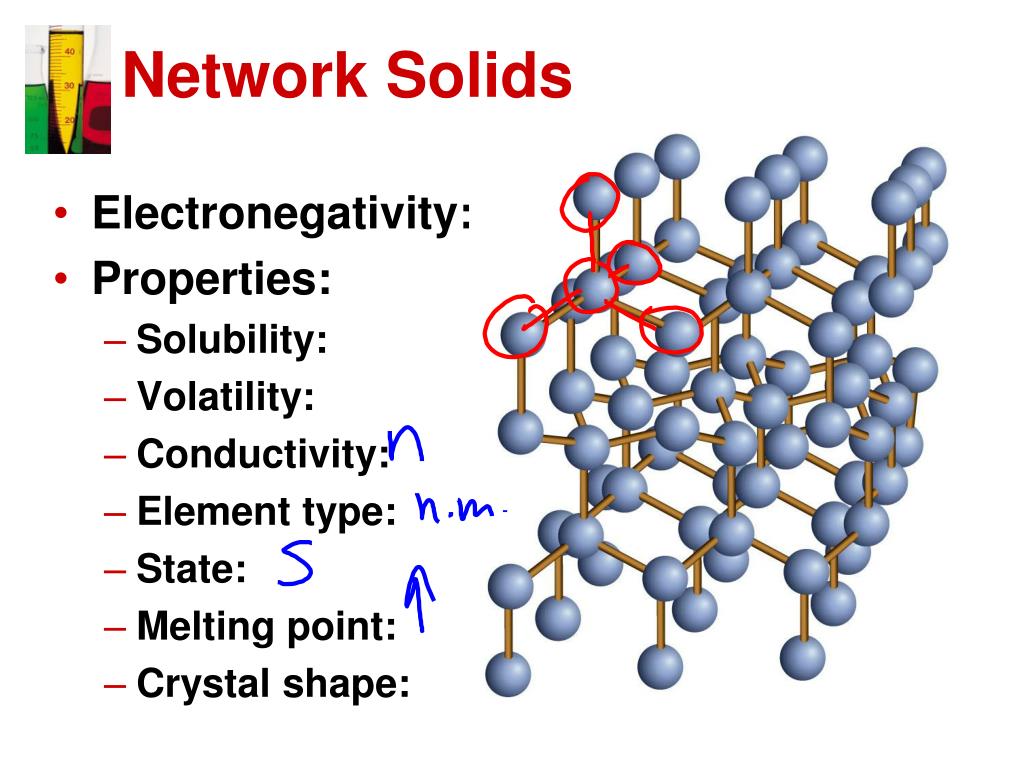
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
